From audit to shipped design system — rethinking the UX of a mission-critical pharma lab instrument.
The Growth Direct System automates microbial quality control testing in pharmaceutical manufacturing — the kind of environment where a contamination event can halt production, compromise patient safety, and trigger regulatory action. The software running these labs had to meet FDA's 21 CFR Part 11 compliance requirements: every action logged, every decision traceable.
I joined Rapid Micro Biosystems to redesign RMBNucleus™ — the web-based admin interface and the embedded touchscreen on the instrument itself. The existing system had been built by engineers without a dedicated designer. I started with a full UX audit, ran usability testing on internal staff, and worked through wireframes before building a component library that covered both interfaces.
My Role
Product Designer
Timeline
2024–2026 (1 yr 4 mo)
Team
Backend and frontend engineers, project manager
Responsibilities
UX audit, wireframing, usability testing, UI design, design system
Result
Scale 150+ Growth Direct systems deployed
The challenge
The Growth Direct System runs continuously in pharmaceutical labs. Operators load cassettes, monitor incubators, and approve test results, often under time pressure, in environments where mistakes have real consequences. Administrators configure the system remotely through a web interface, managing users, test parameters, and compliance reporting.
Two distinct users. Two distinct interfaces. One regulatory environment where every interaction needs to be deliberate, logged, and defensible.
The existing software had been designed by engineers. It worked, but it put the burden of navigation on the user. Running a single test required assembling three separate configuration components: a Method, an Action Alert Level, and a Handling Rule. There was no way to save that combination as a template. No search. No filtering across long lists. Every session started from scratch.
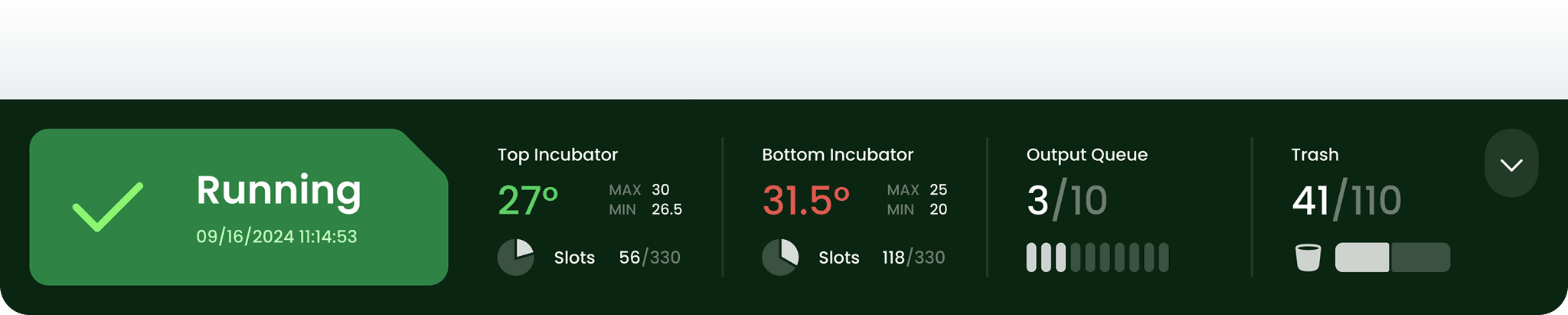
Device status bar
The embedded touchscreen on the Growth Direct instrument has a permanent status bar at the bottom of every screen. It is always visible, regardless of what the operator is doing on the device.
The left side of the bar holds the most important element: the system status indicator. It is intentionally large — designed so that an operator can read the system state by color from across the room, without walking up to the device. Green means running. Any deviation is immediately visible at a distance.
The rest of the bar shows the four key operational metrics: incubator temperature for both chambers, available cassette slots, output queue fill level, and trash bin capacity. All four require human intervention when they reach a threshold — temperature out of range, no slots left, output queue full, trash bin full. The bar signals each state directly, without requiring navigation.
When an operator needs more screen space to review data, the status bar can be minimized. The essential indicators collapse but remain accessible — the system state is never fully hidden.
In a system designed to run unattended for extended periods, this persistent visibility is not a convenience. It is the first line of awareness for everyone in the lab.
Outcome
The redesigned system shipped as RMBNucleus™ — deployed across Rapid Micro Biosystems' global install base of Growth Direct instruments, used in pharmaceutical manufacturing facilities across the US, Europe, and Japan.
Usability testing confirmed that navigation became clearer and test configuration faster. The Sample template system eliminated a recurring source of configuration friction. Internal staff reported that onboarding new operators on the redesigned system required less time than the previous version.